Listen to our webinar on the PFAS family of compounds held April 9, 2019. It covers:
PFAS in likely and unlikely places
Investigation and remediation
The future of PFAS regulations and cleanup standards
Per- and polyfluoroalkyl substances (PFAS) are a family of compounds historically used as water and stain repellants, fire suppressants and nonstick coatings, among other uses. Emerging research into the toxicology and chemical fate and transport of these compounds is raising serious red flags by scientists and regulators.
Many state and federal agency websites indicate that the PFAS family includes about 3,000 individual manufactured chemicals; however, some experts believe that the actual number of PFAS compounds may exceed many times that amount if environmental intermediates (i.e., compounds produced as the original chemicals transform in the environment) are included in the total.
As shown in the figure, non-polymer PFAS can broadly be segregated into two groupings: perfluoroalkyl substances and polyfluoroalkyl substances.
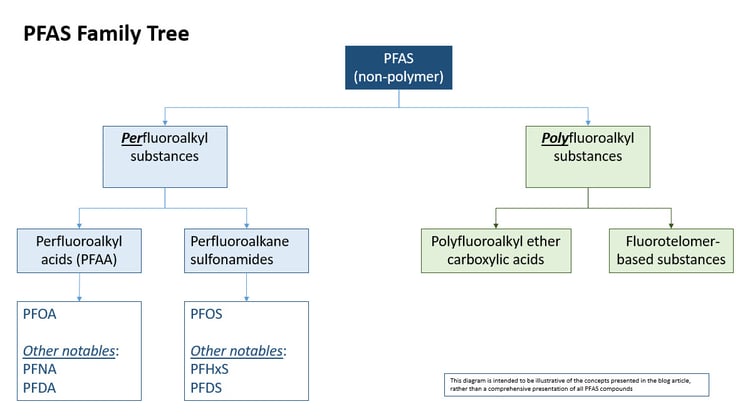
Both groupings include compounds where a chain of carbon atoms (this is the “alkyl” part) is attached to a functional group (changing the functional group is one of the primary ways the individual compounds differ from one another). In alkyl hydrocarbons (such as propane and butane), hydrogen atoms are attached to the carbon chain. In perfluoroalkyl substances, all of the hydrogen atoms are replaced by fluorine atoms, except for that functional group part. Polyfluoroalkyl substances are similar, except that only some of the hydrogen atoms are replaced by fluorine atoms (again, except for the functional group).
.jpg?width=1920&name=shutterstock_172594652%20(2).jpg)
The fluorinated part of the molecule is what makes this class of substances effective as stain repellant or nonstick coatings, as well as in fire suppressants. Generally, the functional group makes the molecules more water-soluble than they “should be” given the rest of their structure. These functional groups further segregate compounds within the PFAS family tree, but we can leave that for another article.
It’s generally accepted that the perfluorinated PFAS compounds (which include PFOA and PFOS) are more toxic than polyfluorinated PFAS compounds. Consequently, states are largely focused on setting cleanup objectives for perfluorinated PFAS compounds. For example, the best-known PFAS compounds, perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) are the most consistently included compounds in lists of PFAS cleanup objectives and action levels.
Generally accurate, but not so with PFAS. It’s true that the action levels for PFAS compounds are extraordinarily low due to their toxicity. These action levels are comparable to other known classes of highly toxic compounds, such as polychlorinated biphenyl (PCBs) and dioxin. PCBs and dioxin are very insoluble and, as a result, are generally immobile in the environment; however, PFAS are relatively soluble by comparison because of those functional groups.
As a result, PFAS compounds are capable of migrating widely via groundwater, unlike other similarly toxic compounds. Interestingly, a primary long-term groundwater contaminant source of PFAS in the subsurface is the vadose zone. (More on that in a future paper.) All of this makes PFAS fairly unique for remediation practitioners and makes investigations and risk evaluations complex.

Currently, there are no federal cleanup standards for PFAS in soil or groundwater. States are filling that gap, albeit in an inconsistent way. State cleanup objectives are largely focused on perfluorinated PFAS compounds, as they are known to be the most toxic compounds in the PFAS family. However, as researchers learn more about the transport and fate of PFAS compounds in the environment, a broader suite of PFAS may become important in site investigations.
.jpg?width=1920&name=shutterstock_1129566662%20(2).jpg)
Yes and no. With so many compounds in the PFAS family, it’s unreasonable to set standards for all of them, in which case setting standards for the most studied, most toxic compounds makes sense. In some ways, this is analogous to setting standards for benzene, toluene, ethylbenzene, and xylenes at gas station sites when gasoline is really a soup of hydrocarbons. However, in the environment and in organisms, polyfluorinated PFAS compounds are known to oxidize into perfluorinated PFAS compounds under the right conditions.
Consequently, investigating only for perfluorinated PFAS compounds may result in data that suggests that no problem is present if only polychlorinated PFAS are present in the sample locations. If those polyfluorinated compounds oxidize into perfluorinated compounds, the data from that initial investigation can result in a “false negative” regarding risk.
Need help with a PFAS site or project? Contact our experts.
Tim Bradburne, P.G., Principal
Scott Warner, P.G., C.H.G., C.E.G., Principal Hydrogeologist
Listen to our webinar on the PFAS family of compounds held April 9, 2019. It covers:
PFAS in likely and unlikely places
Investigation and remediation
The future of PFAS regulations and cleanup standards
140 South Dearborn Street, Suite 1520, Chicago, IL 60603
800-875-1756